FL6000Dual modulation chlorophyll fluorescence measuring instrument

FL6000The dual modulation chlorophyll fluorescence analyzer is the latest upgraded version of the FL3500 dual modulation chlorophyll fluorescence analyzer, specifically designed as a powerful scientific tool for in-depth research on the photosynthesis mechanism of microalgae, chloroplasts, or thylakoid suspensions such as blue-green algae or green algae. The instrument has dual channel measurement control, which can control the temperature of the measured sample, and is equipped with single flip light (STF). It has multiple measurement programs that can be modified by users, and can conduct in-depth research on various mechanisms of chlorophyll fluorescence internationally. Its core structure includes an optical measuring head with a suspension standard sample cup, built-in 3 sets of LED light sources, and 1 1MHz/16 bit AD converted PIN diode signal detector. The gain and integration time of AD conversion can be controlled by software. The detector can measure chlorophyll fluorescence signals with a time resolution of up to 4 µ s (1 µ s for the fast version).
Application fields:
·Screening of plant photosynthetic characteristics and metabolic disorders
·Detection of biotic and abiotic stress
·Research on Plant Stress Resistance or Susceptibility
·Research on metabolic disorders
·Research on the Working Mechanism of Photosynthetic Systems
·Research on Photosynthetic Physiological Response Strategies of Stressed Plants

Typical sample:
·Blue algae (cyanobacteria)
·green algae
·Chloroplast suspension
·Thylakoid suspension
·plant debris
Features:
·Built in chlorophyll fluorescence induction measurement, PAM (pulse modulation) measurement, OJIP rapid fluorescence kinetics measurement, QA - re oxidation kinetics, S-state transition, chlorophyll fluorescence quenching and other measurement programs, it is widely recognized as the most comprehensive chlorophyll fluorescence meter in the world
·Dual modulation technology, capable of dual color modulation of measurement light, with modulation of photochemical light and sustained photochemical light, can perform STF (single turn flash), TTF (double turn flash), MTF (multi turn flash) and customized FRR technology (Fast Repetition Rate) measurements
·The standard version has a time resolution of 4 µ s, while the fast versionUp to 1 µ s, it is currently the chlorophyll fluorescence analyzer with the highest time resolution
·The control unit is dual channel and can be connected to temperature sensors for temperature control and oxygen measurement units for Hill reaction measurement
·Has extremely high sensitivity, with a minimum detection limit of 100ng Chla/L
·Measurement light, actinic light, saturated DSLR converted light source color and intensity can be customized
·The host is equipped with a color touch screen display, which allows real-time viewing of fluorescence curve graphs
Technical Specifications:
·Experimental procedure: Measurement of Kautsky chlorophyll fluorescence induction effect; PAM (Pulse Modulation)Fluorescence quenching kineticsMeasurement; OJIP rapid fluorescence kinetics measurement; QA - Redox kinetics; S state transition; Rapid chlorophyll fluorescence induction
Fluorescence parameters:
U SPAMFluorescence quenching kinetics measurement: Measure the fluorescence quenching kinetics curve and calculate F0,Fm,Fv,F0’,Fm’,Fv’,QY(II),NPQ,ΦPSII,Fv/Fm,Fv’/Fm’,Rfd,qN,qP,ETRWaiting for over 50 chlorophyll fluorescence parameters;
U SOJIPRapid Fluorescence Dynamics Measurement: Measure the OJIP rapid fluorescence kinetics curve, which can calculate F0、FJ、Fi、Fm、Fv、VJ、Vi、Fm / F0、Fv / F0、Fv / Fm、M0、Area、Fix Area、SM、SS、N、Phi_P0、Psi_0、Phi_E0、Phi_D0、Phi_Pav、ABS / RC、TR0/ RC、ET0/ RC、DI0/ RCWaiting for more than 20 related parameters;
U SQAQA reoxygenation kinetics: Measure the QA reoxygenation kinetics curve to fit the amplitudes (A1, A2, A3) and time constants (T1, T2, T3) of the fast phase, middle phase, and slow phase during the QA reoxygenation process
U SSState transition (S-state test): Measure the fluorescence attenuation curve of S-state test, used to fit and calculate the non active photosystem II (PSII)X)Number of reaction centers
U SFlash Fluorescence Induction (FFL, fast version only): used for fitting and calculating effective antenna area, antenna connectivity, etc
U SProvide user-defined protocol functionality to achieve PSII antenna heterogeneityalphaWith PSIIBAnalysis and PSII effective antenna cross-sectional area(sPSII)Measurement of parameters (optional customization function)
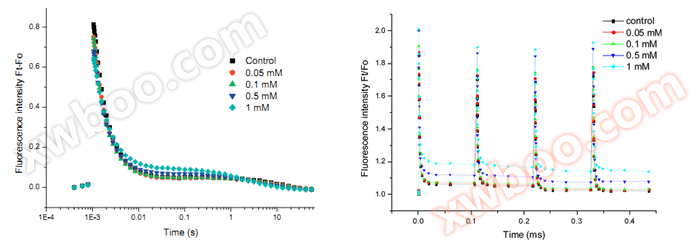
U SQA- Redox kinetics curve andS-state testFluorescence attenuation curve(Li,2010)
·Time resolution (sampling frequency): High sensitivity detector, standard version has a time resolution of 4 µ s, fast version has a time resolution of 1 µ s
·Minimum detection limit: Standard version 100ng Chla/L, Rapid version 1 μ g Chla/L
·Control unit: equipped with a color touch screen display, allowing real-time viewing of fluorescence curve graphs

·Measurement Room:
oMeasurement of flicker: 623nm red orange light and 460nm blue light, with a flicker time of 2-5 µ s
oSingle cycle saturation flash: maximum light intensity of 170000 µ mol (photons)/m ². s, flash time of 20-50 µ s
oContinuous photochemical light: maximum light intensity of 3500 µ mol (photons)/m ². s
oFluorescence detector: PIN photodiode
oADConverter: 16 bit
oSample test tube: bottom area of 10 × 10mm, volume of 4ml
Customized measurement room (optional): can customize measurement light, saturation flicker, photochemical light colors (blue, cyan, amber, etc.) and detection bands (ChlA, ChlB) separately
Far infrared light source (optional): used for measuring F0The wavelength is 730nm
·Oxygen measurement module (optional): measures the oxygen release of algae
·Temperature control (optional): TR 6000 temperature regulator, temperature control range 5-60 ℃, accuracy 0.1 ℃
Electromagnetic stirring (optional): used for sample mixing to prevent sample precipitation, can be manually adjusted or automatically controlled by software
Communication interface: RS232 serial port/USB
FluorWinSoftware: Define or create experimental plans, light source control settings, data output, analysis and processing, and chart display

Typical applications:
1. Researcher Wang Qiang from the Institute of Hydrobiology, Chinese Academy of Sciences demonstrated that nitrite stress first affects the PSII receptor side of Synechocystis sp. PCC 6803 using FL3500 chlorophyll fluorescence analyzer (models before FL6000) and TL plant thermoluminescence system (Zhan X, et al, 2017). The in-depth study of the mechanism of photosynthesis often requires the combination of these two instruments.

2.Pan Zhongliang, a researcher at the Xinjiang Institute of Ecology and Geography of the Chinese Academy of Sciences, and his research team conducted in-depth toxicological studies on harmful substances such as heavy metals, salts, toxic compounds, herbicides, insecticides, and antibiotics on algae using the FL3500 chlorophyll fluorescence analyzer (model before FL6000). Through FL3500's unique high-resolution OJIP rapid fluorescence kinetics measurement, QA re oxidation kinetics, S state transition and other chlorophyll fluorescence measurement programs, the toxic mechanisms and ecological impacts of different concentrations and treatment times on algal photosynthetic systems were comprehensively revealed. At present, Pan Zhongliang's research group has published more than 20 high-level articles in international SCI journals and domestic core journals using FL3500 (models before FL6000).

Origin: Czech Republic
References:
1. Manaa A, et al. 2019. Salinity tolerance of quinoa (Chenopodium quinoaWilld) as assessed by chloroplast ultrastructure and photosynthetic performance. Environmental and Experimental Botany 162: 103-114
2. Yu Z, et al. 2019. Sensitivity of Chlamydomonas reinhardtii to cadmium stress is associated with phototaxis. Environmental Science: Processes & Impacts 21: 1011-1020
3. Liang Y, et al. 2019. Molecular mechanisms of temperature acclimation and adaptation in marine diatoms. The ISME journal, DOI: 10.1038/s41396-019-0441-9
4. Orfanidis S, et al. 2019. Solving Nuisance Cyanobacteria Eutrophication Through Biotechnology. Applied Sciences 9(12): 2566
5. Sicora C I, et al. 2019. Regulation of PSII function inCyanothecesp. ATCC 51142 during a light–dark cycle. Photosynthesis Research 139(1–3): 461–473
6. Smythers A L, et al. 2019. Characterizing the effect of Poast onChlorella vulgaris, a non-target organism. Chemosphere 219: 704-712
7. Albanese P, et al. 2018. Thylakoid proteome modulation in pea plants grown at different irradiances: quantitative proteomic profiling in a non‐model organism aided by transcriptomic data integration. The Plant Journal 96(4): 786-800
8. Antal T, Konyukhov I, Volgusheva A, et al. 2018. Chlorophyll fluorescence induction and relaxation system for the continuous monitoring of photosynthetic capacity in photobioreactors. Physiol Plantarum. DOI: 10.1111/ppl.12693
9. Antal T K, Maslakov A, Yakovleva O V, et al. 2018.Simulation of chlorophyll fluorescence rise and decay kinetics, and P700-related absorbance changes by using a rule-based kinetic Monte-Carlo method. Photosynthesis Research. DOI:10.1007/s11120-018-0564-2
10.Biswas S, Eaton-Rye J J, et al. 2018. PsbY is required for prevention of photodamage to photosystem II in a PsbM-lacking mutant ofSynechocystissp. PCC 6803. Photosynthetica, 56(1), 200–209.
11.Bonisteel E M, et al. 2018. Strain specific differences in rates of Photosystem II repair in picocyanobacteria correlate to differences in FtsH protein levels and isoform expression patterns. PLoS ONE 13(12): e0209115.
12.Fang X, et al. 2018. Transcriptomic responses of the marine cyanobacteriumProchlorococcusto viral lysis products. Environmental Microbiology, doi: 10.1101/394122.
13.Kuthanová Trsková E, Belgio E, Yeates A M, et al. 2018. Antenna proton sensitivity determines photosynthetic light harvesting strategy, Journal of Experimental Botany 69(18): 4483-4493

